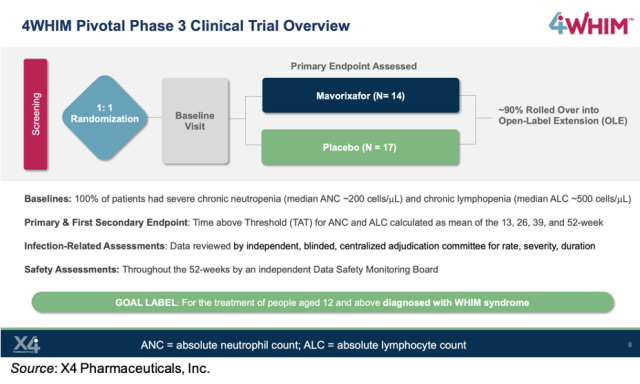
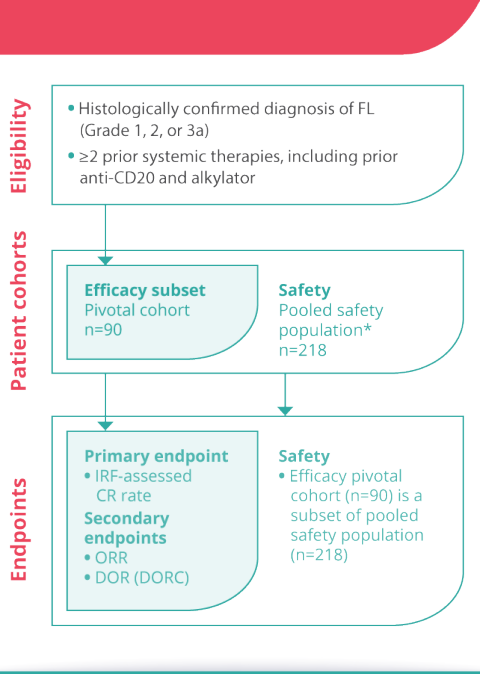
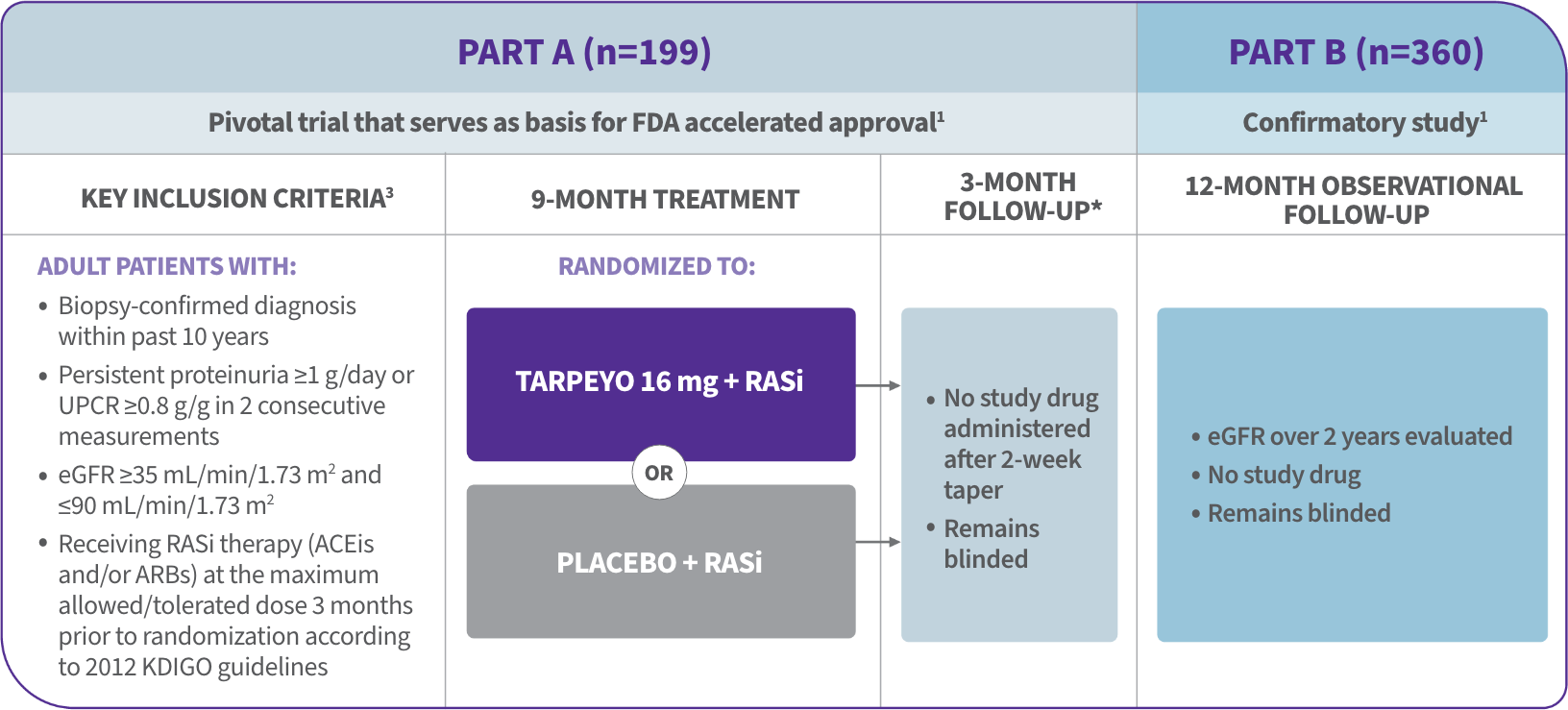
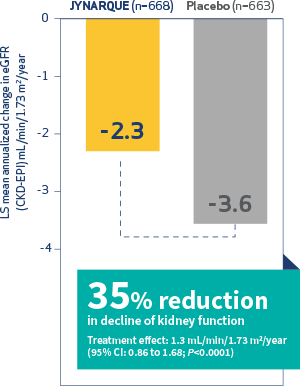
Alexion Announces Planned Initiation of Pivotal Phase 3 Study of ULTOMIRIS® (ravulizumab) in ALS - Clinical Trial Recruitment

Characteristics of Single Pivotal Trials Supporting Regulatory Approvals of Novel Non‐orphan, Non‐oncology Drugs in the European Union and United States from 2012−2016 - Morant - 2019 - Clinical and Translational Science - Wiley Online Library
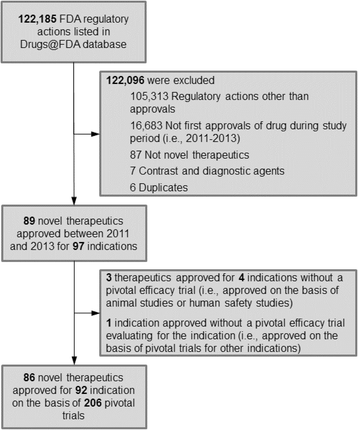
Participation of the elderly, women, and minorities in pivotal trials supporting 2011–2013 U.S. Food and Drug Administration approvals | Trials | Full Text